
777 NW 9th Ave, Suite 404, Corvallis, OR 97330
777 NW 9th Ave, Suite 404, Corvallis, OR 97330
About Us
Mo-99 Background
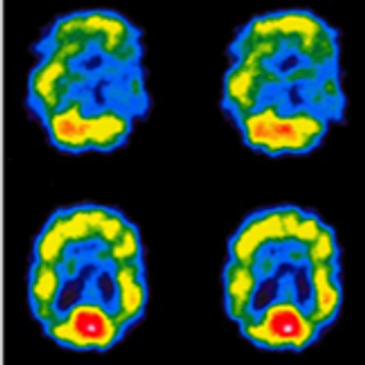
The most common radioisotope used in nuclear medicine procedures in the U.S. is Tc-99m. Tc-99m is used extensively in nuclear cardiology (50% of procedures), nuclear oncology (25%), and in other imaging of the brain, endocrine system, lungs, gastrointestinal, genitourinary, and bones. There are currently 40 million nuclear medicine procedures performed in the U.S. each year, with 80% using Tc-99m based radiopharmaceuticals. Tc-99m is most conveniently obtained from the beta-decay of Mo-99, which has a half-life of
66 hours. Due to the short half-lives of Tc-99m (6 hours) and Mo-99, the isotopes cannot be stockpiled and must be produced in fresh batches and distributed to medical imaging centers almost daily.
Our Company

NWMI is dedicated to providing a reliable, securable, and affordable source of Mo-99 both domestically and worldwide by early 2023. We have embraced this challenge and will safely apply our technical and project expertise to ensure project success. Our people drive our solution!
Management Team

Our highly qualified management team includes experts on complex nuclear EPC projects, nuclear reactor licensing, research reactor operations, and nuclear criticality safety who are focused on designing and building a radioisotope production facility (RPF) to deliver in-specification Mo-99 to our customers.
Our Commitment
We are committed to advancing radioisotope technology development to support human health advancements and improve lives through the use of medical isotope imaging.
Mo-99 Background
The most common radioisotope used in nuclear medicine procedures in the U.S. is Tc-99m. Tc-99m has found extensive use in nuclear cardiology (50% of procedures), nuclear oncology (25%), and in other imaging of the brain, endocrine system, lungs, gastrointestinal, genitourinary, and bones. There are currently 40 million nuclear medicine procedures performed annually in the U.S., with 80% using Tc-99m based radiopharmaceuticals. Although the radioisotope can be produced directly on a cyclotron or other type of particle accelerator, Tc-99m is most conveniently obtained from the beta-decay of Mo-99. Due to the short half-lives of Tc-99m (6 hours) and Mo-99 (66 hours), the isotopes cannot be stockpiled and must be produced in fresh batches and distributed to medical imaging centers almost daily.
Mo-99 is a fission fragment that is abundantly produced in the neutron-induced fission of U-235 (6% of all fissions). The Tc-99m generators allow a quick and convenient chemical separation of Tc-99m daughter nuclei from the Mo-99 parent material. The 66-hour half-life enables Mo-99 to be produced at large-capacity locations and then transported to centralized radiopharmacies in the U.S., which distribute the Tc-99m generators to hospitals and other imaging facilities. Mo-99 production is typically measured in “6-day curies” based on the material activity 6 days after being shipped (the actual value is roughly four times the 6-day value). The estimated worldwide demand for Mo-99 is ~9,400 6-day Ci per week (2018).
The last commercial production of Mo-99 in the U.S. ended in 1989 and the process was just restarted in 2018. Domestic supply has and will continue to rely on international producers who employed the high efficiency of irradiating highly enriched uranium (HEU) targets, with much of the HEU exported from the U.S. Currently, six multipurpose research reactors, with five of these sites being over 45–55 years old, irradiate HEU or low--enriched uranium (LEU) targets for Mo-99 production. As part of nuclear nonproliferation efforts, under the American Medical Isotope Production Act of 2012, the U.S. must stop exporting HEU by 2020, which is used for targets for isotope production and for fuel for reactors. The industry has largely converted to the use of LEU.

Our Company
Northwest Medical Isotopes, LLC:
- Is US-based and funded as a domestic supplier of Mo-99
- Will produce 50% of US market demand weekly
- Has developed a low risk approach to production of Mo-99
- Ensures reliability through use of multiple reactors for irradiation services and redundancy in the separation and purification facility
- Does not require new irradiation technology (accelerators) or new reactors to execute our mission
- Recycles LEU to minimize environmental impact and reduce operating costs
- Offers experienced management and a highly qualified team for designing and building the needed production facility
- Will be able to deliver in-specification Mo-99 to our customers, as needed
- Offers an established Mo-99 separation and purification technology
- Supports the U.S. Government goal of Mo-99 production without the use of highly enriched uranium
- Has the plans, people, assets, and financing in place to achieve the mission
- Is committed to supporting human health and improving lives through the use of medical isotope imaging
Our Mission
Establish and deliver a domestic reliable, securable source of at least 3,000 6-day curies of Mo-99 weekly, steady-state and without the use of highly enriched uranium
Radioisotope Production Facility

RPF Purpose and Production Process
NWMI will design, license, and construct a radioisotope production facility (RPF) to process low-enriched uranium (LEU) targets to produce at least 3,000 6‑day Ci of Mo-99 through a fission-based process. The RPF will house the entire production process, which includes:
- Receiving LEU from U.S. Department of Energy (DOE)
- Producing LEU target materials and fabrication of targets
- Packaging and shipping LEU targets to the university reactor network for irradiation
- Returning irradiated LEU targets for dissolution, recovery, and purification of Mo-99
- Packaging Mo-99 product to prepare for transport to radiopharmaceutical generator manufacturers
- Recovering and recycling LEU to minimize radioactive, mixed, and hazardous waste generation
- Treating/packaging wastes generated by RPF process steps and prepare for transport and disposal
The targets will be chemically processed to produce pharmaceutical grade Mo-99. The finished product will be produced using the current established Good Manufacturing Practices program. The process used will also meet FDA requirements applicable to active pharmaceutical ingredients.

RPF Site
The RPF site will be situated within Discovery Ridge Research Park (Discovery Ridge) in Columbia, Missouri. The site is in central Missouri, approximately 125 miles east of Kansas City and 125 miles west of St. Louis. The Missouri River lies 9.5 miles west of the site. The RPF site is located 3.5 mi southeast of the main University of Missouri (MU) campus and occupies 15 acres (Lots 14 and 15) at Discovery Ridge on property owned by MU.
The layout of the NWMI site, including the RPF, is shown at right. Three adjacent, separate buildings will be located on the site, including the Administrative Building (outside of the protected area), Waste Staging and Shipping Building for additional Class A waste storage (inside the protected area), and Diesel Generator Building. These major facilities also receive, store/hold, or process chemicals, oil, diesel fuel, and other hazardous and radioactive materials.
Reactor Fission – Gold Standard
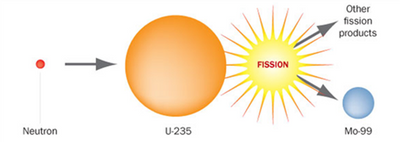
NWMI is producing Mo-99 by reactor fission (splitting a uranium atom).” Uranium fission is considered to be the “gold standard” process for producing Mo-99, as the production process is highly efficient and the Mo-99 produced has a high specific activity (>1,000 Ci/g, making it suitable for use in conventional Tc-99m generators used worldwide.
Typically, targets containing U-235 are placed in a nuclear reactor for about 5 to 7 days to allow Mo-99 to build up to between about 70% and 80% of saturation concentration. The neutrons given off by the nuclear reactor split the U-235 atoms in the targets, continuously creating Mo-99 at a known rate. Whenever the U-235 is split, 6% of the time, one of the fragments created is Mo‑99. Approximately 3% of the U-235 in the target is consumed during irradiation. The targets are removed from the reactor after irradiation and a 24‑hour (approximately) cool-down period is completed.
Progress To Date
NWMI has completed the following activities to date:
Radioisotope Production Facility
- Received NRC approval for RPF construction (May 2018, NRC CLI‑18‑06); final EIS published by NRC (May 2017, NUREG‑2209)
- Received approximately $7M in tax abatements from Boone County, Missouri (September 2018)
- Awarded $30M 50/50 cost share Cooperative Agreement from DOE NNSA, which will support RPF final design and NRC Operating License Application development and submission (August 2019)
- Finalized RPF siting and land lease (15 acres) at Discovery Ridge Research Park in Columbia, Missouri near University of Missouri Research Reactor (MURR) (November 2019)
- Initiated development of NRC Operating License Application (estimated completion September 2020)
- Initiated RPF final design (estimated completion September 2020)
Research and development (R&D)
- Completed cold and hot small-scale irradiation and processing demonstration tests (2019)
- Completed and continuing large-scale Mo-99 production to support Tc-99m generator testing (2019)
- Completed target material production with both natural uranium and LEU (2019)
- Performing demonstration activities, including optimization of process technologies and equipment and scaling of processes to commercial quantities (ongoing)
University research reactors
- University reactor network identified (MURR and Oregon State University TRIGA Reactor [OSTR]); commercial irradiation services agreements complete (January 2019)
- OSTR license amendment approved by the NRC for irradiation of up to three prototypic targets (January 2016)

Management Team

Carolyn Haass, Chief Operating Officer
Ms. Haass has 36 years of experience on complex nuclear, chemical, hazardous, and mixed-waste EPC projects for government and private industry. She has extensive communications experience in the nuclear and environmental industry, including day-to-day interface with regulators, safety boards, Congress, stakeholders, tribal nations, public, media, community leaders, and decision makers. Ms. Haass has over 10 years of medical isotope production experience and is responsible for development, coordination, and submission to the NRC of the 10CFR50/70 Operating License Application and safety analysis report in support of RPF operations. Under her direction, the RPF Construction Permit Application was submitted to and approved by the NRC in 2018 for the construction of the RPF under 10CFR50.

Ralph Butler, PE, Senior Technical Advisor
Mr. Butler has over 36 years of nuclear experience in the management, oversight, and operation of Navy, commercial, university, and DOE facilities, both domestic and international. His experience includes power plant and research reactor operations, program management, quality improvement and assessment activities, and independent safety reviews. He is a Registered Professional Engineer in the state of Missouri and held an NRC Senior Reactor Operator License for Westinghouse Pressurized Water Reactor, Callaway Nuclear Energy Center. He serves as Senior Technical Advisor on the NWMI RFP project and as NWMI's liaison with MURR.

Mike Corum, Engineering Manager
Mr. Corum is a subject matter expert with over 28 years of nuclear industry experience, including 9 years of medical isotope production experience. His broad experience base in nuclear criticality safety includes expertise in radiation shielding, hazards analysis, packaging and transportation, and core reload analysis. He has extensive experience in both facility support and packaging and transportation (fresh and spent fuel). Additional areas of expertise include integrated safety analysis, fault tree analysis, qualitative and quantitative methods of hazards assessment, shielding analysis using MCNP code, validation and benchmarking of criticality codes, nuclear reactor core design, and reactor physics calculations in support of reload safety analysis efforts.

Steve Reese, PhD, Irradiation Services Director
Dr. Reese, Director for the OSU Radiation Center, has 22 years of experience in research reactor operations and has a Senior Reactor Operating license for OSTR. The Center is a multifaceted research facility specializing in research related to the nuclear sciences and houses unique capabilities, including the 1.1 MW OSTR, gamma irradiator, thermal hydraulics testing laboratories, and radiochemistry laboratories. Dr. Reese has been a key contributor to the R&D associated with development and testing of the NWMI RPF targets and in supporting the RPF license application process.

Gary Dunford, Process Engineering Manager
Mr. Dunford’s has over 40 years of experience in plant operations and providing process engineering support to radioactive/mixed waste facilities and on medical isotope production facilities. His experience extends to systems engineering applications, flowsheet development, conduct of operations principles, operational readiness activities, safety documentation preparation, and authorization basis implementation. For the past 10 years, Mr. Dunford has supported NWMI facility design of the RPF process systems and development of the RPF license applications and safety analysis.

Ryan Molen, LEED AP BD+C, Construction Project Director
Mr. Molen leads McCarthy’s Advanced Technology & Manufacturing business unit. He has successfully delivered complex laboratory and manufacturing construction projects and nuclear-grade facilities to public entities such as DOE, Oak Ridge National Laboratory, General Services Administration, Department of Veteran’s Affairs, and numerous private clients. He will direct pre-construction and construction activities for the NWMI RPF and associated support facilities.

Artha Petermann, Technical Communications Manager
Ms. Petermann is a Technical Communications specialist, with over 38 years of experience working on technical and cost proposals for various industries and clients, and special projects in the areas of nuclear waste management/cleanup, transportation systems, and computer systems development. Her areas of expertise include proposal development, technical writing and editing, graphics design and editing, promotional materials design and production, and computer systems development and implementation. She has supported NWMI communication activities since 2013 and played a key role in the development of the RPF Construction Permit Application and associated documentation submitted to and approved by the NRC in 2018.
Our Sponsors
Northwest Medical Isotopes, LLC had searched the companies interested in isotopes and found loyal sponsors ready to support our scientific activities.
Alley Cat Allies
Alley Cat Allies is a national advocacy organization dedicated to protecting and improving cats' lives. The NGO actively advocates for Trap-Neuter-Return (TNR), spaying and neutering, anti-cruelty legislation, and for the millions of cats who are euthanized every year in shelters nationwide. Even though the company provides support for the same organizations, its small help for our company matters a lot.
Electronic Frontier Foundation
The Electronic Frontier Foundation champions civil liberties in the digital world. Founded in 1990, EFF champions user privacy, free expression, and innovation through impact litigation, policy analysis, grassroots activism, and technology development. They work to ensure that rights and freedoms are enhanced and protected as our use of technology grows. Northwest Medical Isotopes, LLC expresses its gratitude to this foundation.
Global Zero
Global Zero is an international movement of more than 300 world leaders and half a million citizens worldwide whose mission is to stop the spread of nuclear weapons, secure all nuclear materials, and eliminate all nuclear weapons. The movement engages in direct dialogue with governments and the global community through online and grassroots campaigning.
Random Act of Kindness Foundation
The Random Acts of Kindness Foundation works towards building a kinder world. The organization provides lesson plans to teach kindness in schools and uses the power of the internet to make kindness effortless and to inspire random acts of kindness. In addition, the organization organizes the annual Random Act of Kindness Week.
15M Finance
15M Finance is a connection service focused on helping middle-income, credit-challenged consumers build a better financial path. The company can help its customers with all kinds of loan products: emergency loans and installment loans are its specialties. Also, they offer payday loans. No matter the customer's needs, 15M Finance wants to ensure people have access to the best possible product. And all these without having to worry about whether or not customers will be able to get approved in the first place. 15M Finance works hard daily to ensure people's money isn't just sitting around doing nothing.
Our Commitment
NWMI is an emerging leader with new solutions in nuclear medicine technology:
- Our people drive our solution and are committed to advancing radioisotope technology development to support human health advancements and improve lives through the use of medical isotope imaging.
- We are committed both financially and technically to produce a reliable, securable, and affordable Mo-99 domestically by early 2023
- Environmental Safety, Health, and Quality is our #1 priority
- We are committed to reducing the U.S. healthcare system’s reliance on the fragile, foreign-produced medical isotope supply
- We are committed to ensuring that our processes and products are environmentally friendly.
Career Opportunities

Current Position Openings
NWMI currently has no open positions or job postings. Check back on this webpage for future job postings in the Columbia, Missouri area.
Career Fairs
NWMI will participate in local career fairs when hiring begins to ramp-up. Dates, times, and locations will be posted on this webpage.
Benefits
NWMI will offer a competitive benefits package to full-time NWMI employees. Details will be provided during the interview process.
Employment Application and Resumes
- An employment application will be posted on this webpage when open positions are identified.
- NWMI is not accepting resumes at this time.
Contact Us
Northwest Medical Isotopes, LLC
777 NW 9th Ave, Suite 404, Corvallis, OR 97330